Addiction Neuroscience at TAMU
Research Interests
Our research is focused on the behavioral neuroscience of drug addiction. Addiction is a chronic, relapsing brain disease with high costs to individuals and society. Our research is aimed at a better understanding of the neurobiology underlying the disease so that better treatment and prevention strategies can be developed.
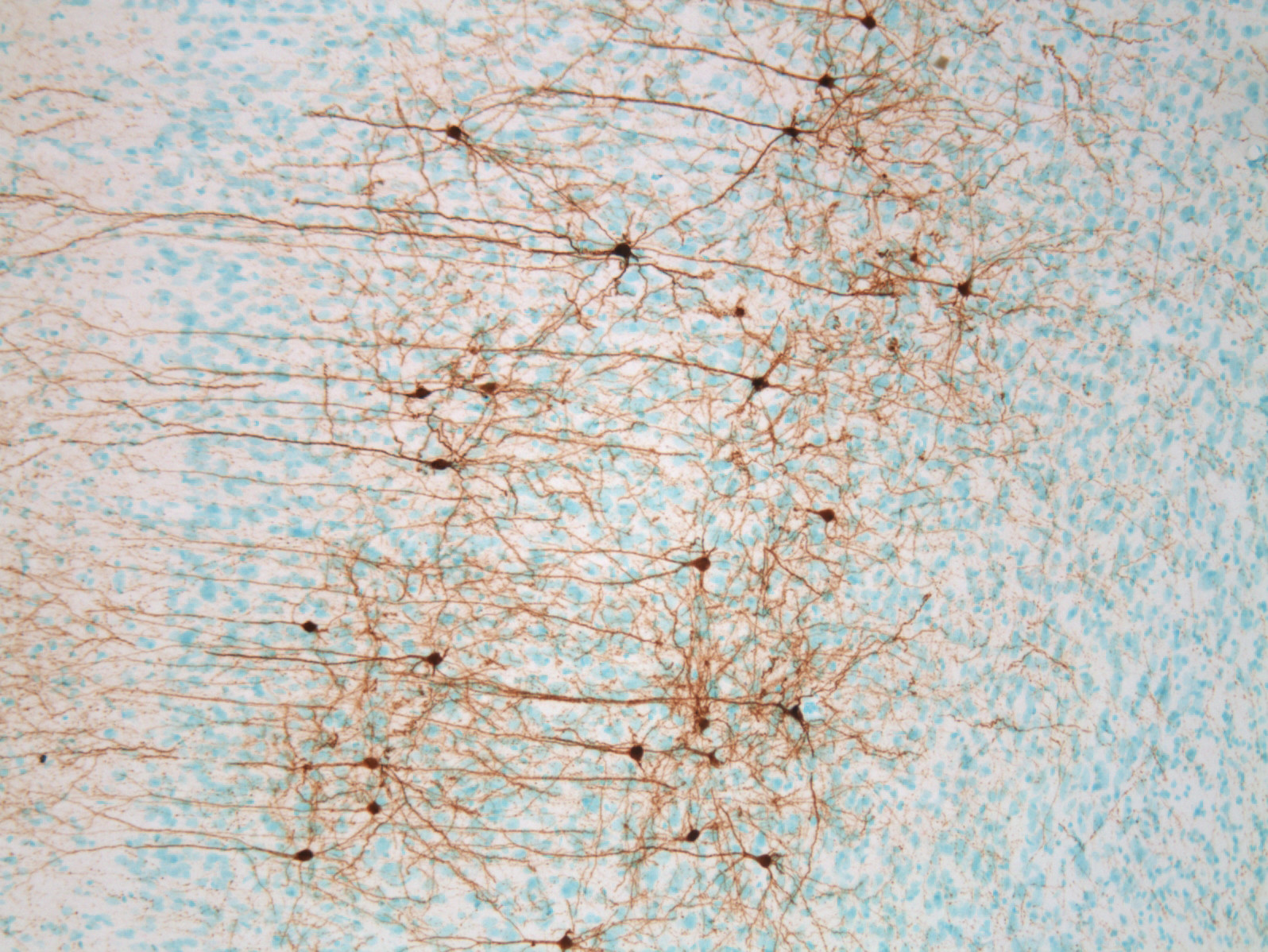
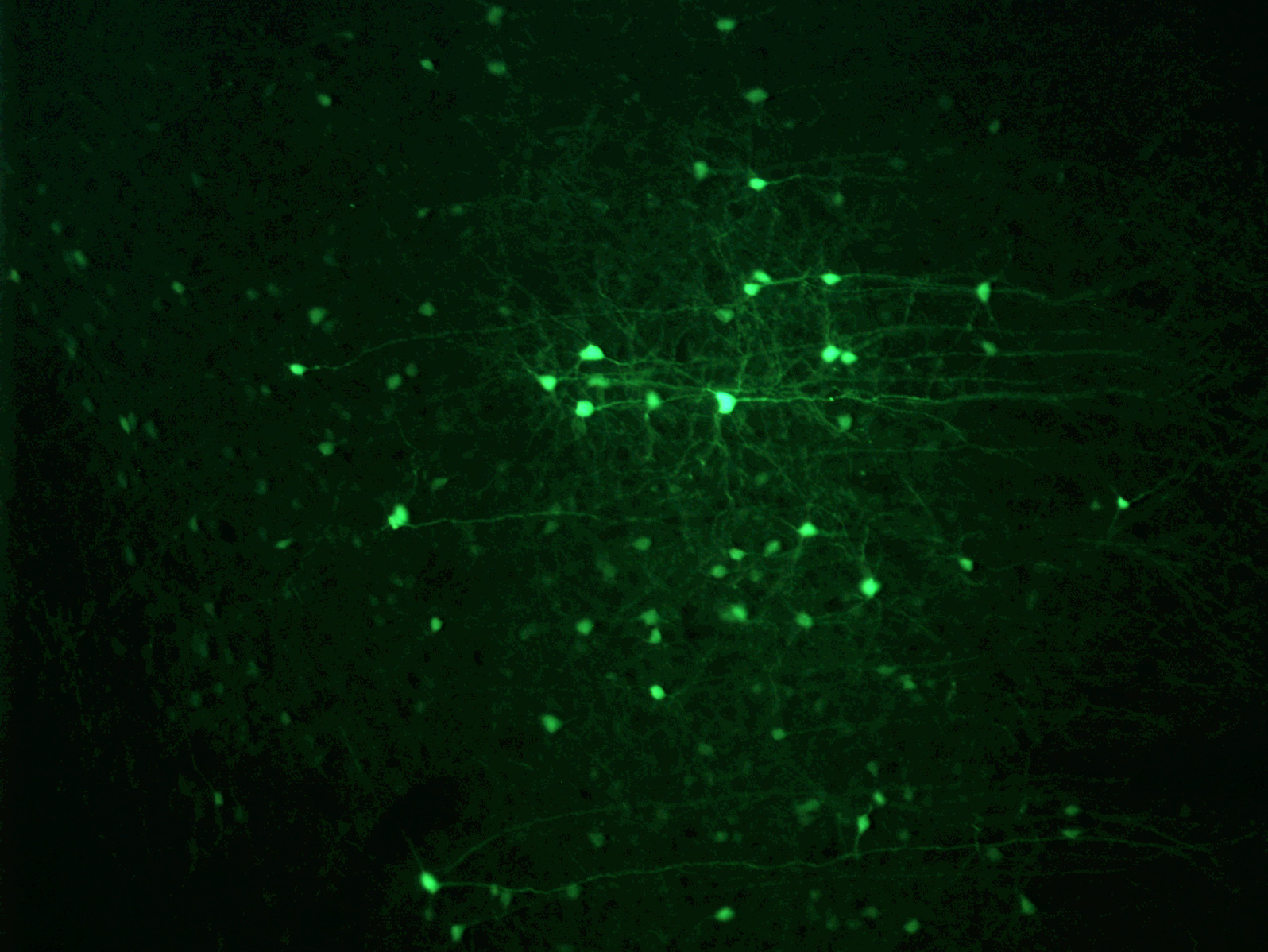
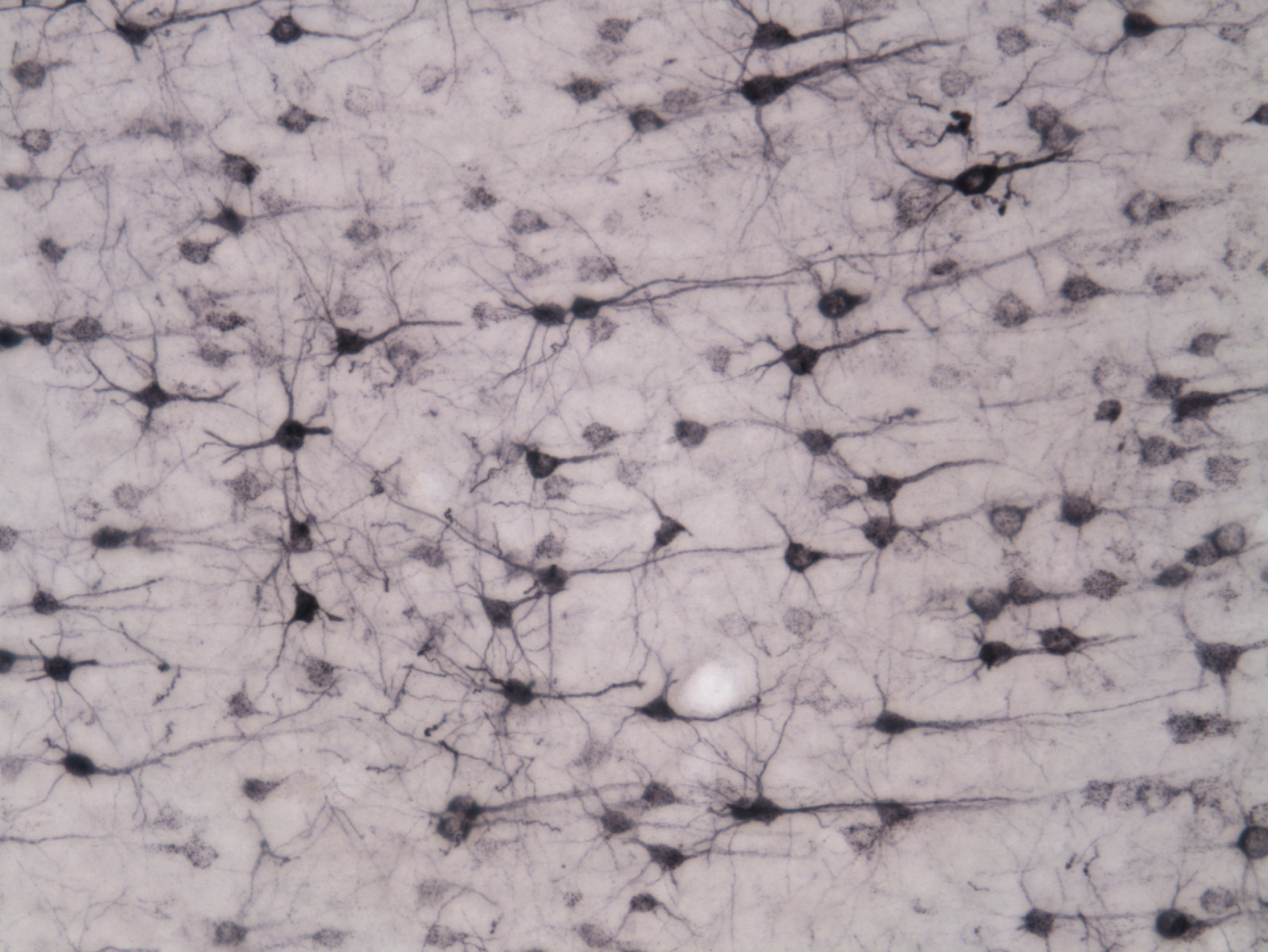
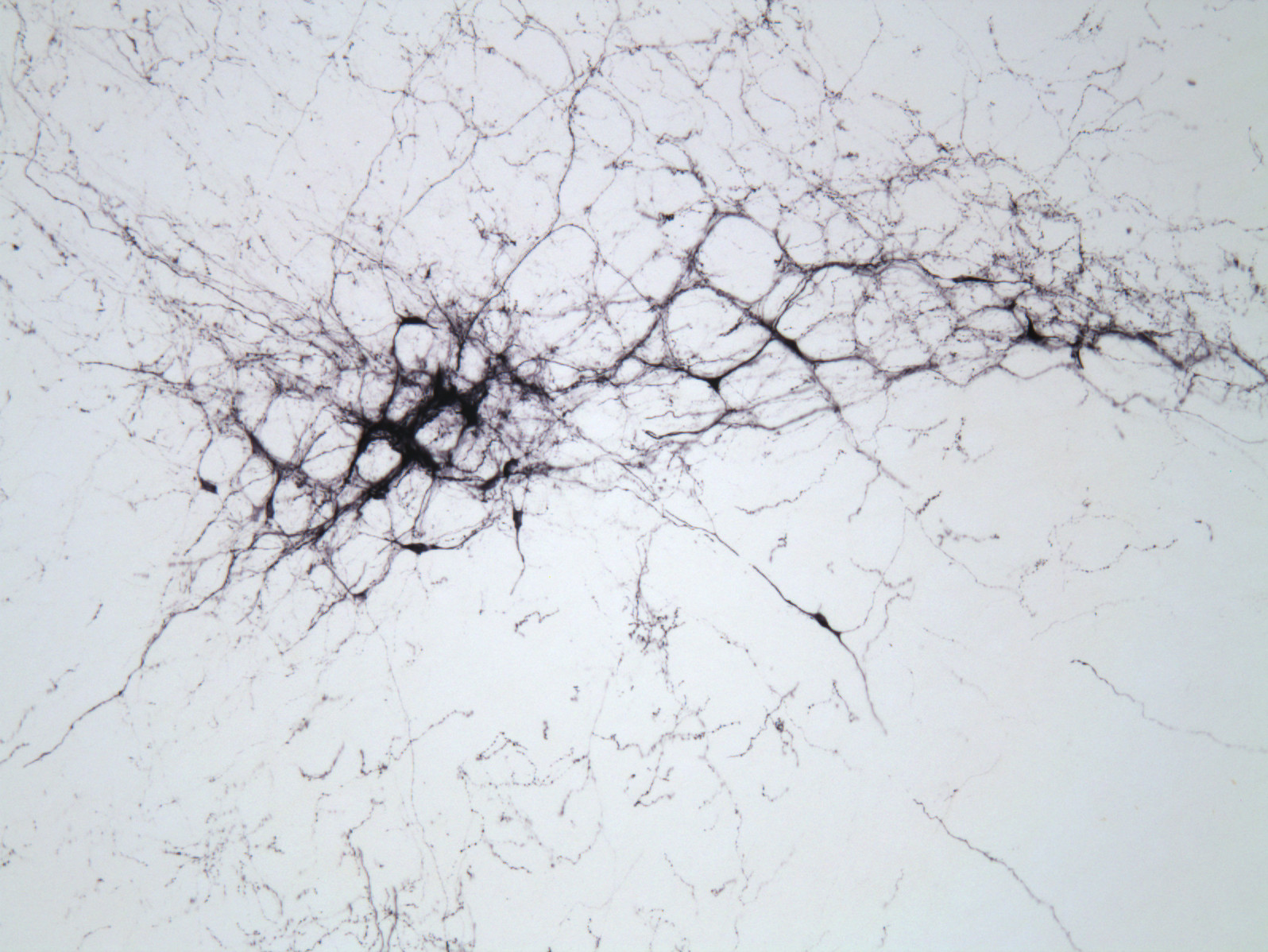
Our work incorporates cutting-edge tools in neuroscience (optogenetics, DREADDs) and classic methodology (pharmacology, lesions, immunohistochemistry, anatomical tracing), with a heavy emphasis on rodent behavioral models such as the self-administration paradigm.